Diseases
(Click on photo to enlarge)
General Disease Management in Cruciferous Weeds
- Crucifer Disease Guide (pdf) – A Practical Guide for Seedsmen, Growers and Agricultural Advisors. Published by Seminis Vegetable Seeds, Inc.’s Plant Health Department and Seed Health Departments.
Diseases
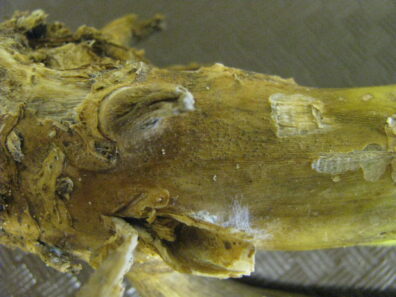
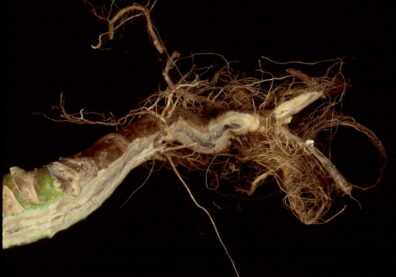
Black Leg
Disease: Black leg
Pathogens: Phoma lingam (sexual stage = Leptosphaeria maculans)
Host crops: Most members of the Brassicaceae (Cruciferae) = cabbage family, including broccoli, Brussels sprouts, cabbage, canola, cauliflower, various Chinese brassica vegetables, collard, kale, mizuna, mustard, oilseed rape, oilseed turnip rape, rutabaga, turnip, etc.), Sinapis (white and yellow mustard), and Raphanus (daikon and radish). Several wild species exist that may be infected by P. lingam including Descurainia (tansymustard), Sisymbrium (hedge mustard), and Thlaspi (penny-cress). This is a quarantine disease in six counties in northwestern WA and all counties east of the Cascade Mountains because of the risk of this pathogen to the brassica vegetable seed industry.

Photo Source: Lindsey du Toit, Washington State University
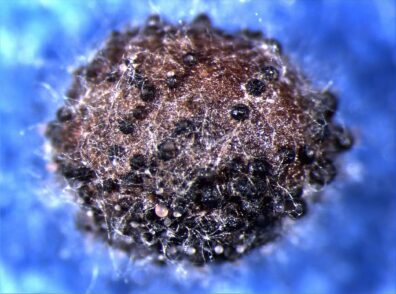
Photo Source: Lindsey du Toit, Washington State University
On-Line Resources:
- Video: Blackleg Disease and Resistance Management. Published by the Canola Council of Canada.
- Pacific Northwest Plant Disease Management Handbook: Cabbage and Cauliflower (Brassica sp.)-Black Leg and Phoma Root Rot
- Pacific Northwest Plant Disease Management Handbook: Seed Crop, Crucifers (Brassica and Raphanus spp.)-Black Leg.
- Black leg in Brassicaceae crops and wild crucifers: 2014 outbreak in the Willamette Valley of Oregon (pdf).
- Black Leg, Light Leaf Spot, and White Leaf Spot (pdf), Cynthia Ocamb, PhD., Plant Pathologist, OSU Extension, Associate Professor–Botany & Plant Pathology.
- Fungicides for Control of Black Leg (pdf), David Priebe, Pesticides Program, Oregon Department of Agriculture.
Addressing Blackleg in the Willamette Valley: Oregon Department of Agriculture permanent ruling released on black leg of brassicaceae in January 2015 – see the Brassica Production Districts document, and the OSDA Permanent Ruling document titled ‘Crucifer blackleg disease requirements moved into one regulation; removes same requirements from rapeseed production districts,’ below.
- Oregon Secretary of State Certificate and Order for Filing (pdf) – PERMANENT ADMINISTRATIVE RULES: Crucifer blackleg disease requirements moved into one regulation; removes same requirements from rapeseed production districts.
- Brassicaceae Production Districts and Rapeseed Control Areas (pdf) (603-052-0860), Oregon Department of Agriculture.
- Management of Black Leg in Oregon on Brassica seed crops (pdf), a Clinic Close-up, Oregon State University Extension Service.
- Management of Black Leg in Oregon on Vegetable Brassica Crops and Seed Crops (pdf), a Clinic Close-up, Oregon State University Extension Service.
Black rot
Disease: Black rot
Pathogens: Xanthomonas campestris pv. campestris
Host crops: Most members of the Brassicaceae (Cruciferae) = cabbage family, including broccoli, Brussels sprouts, cabbage, canola, cauliflower, various Chinese brassica vegetables, collard, kale, mizuna, mustard, oilseed rape, oilseed turnip rape, rutabaga, turnip, etc.), Sinapis (white and yellow mustard), and Raphanus (daikon and radish). Most wild species can be infected by this pathogen. This is a quarantine disease in parts of six counties in northwestern Washington because of the risk of this pathogen to the brassica vegetable seed industry.
Online Resources:
- Cabbage and Cauliflower (Brassica sp.)-Black Rot, Pacific Northwest Handbooks, a Pacific Northwest Extension Publication.
- Black Rot of Crucifers, Fact Sheet, Cooperative Extension, New York State, Cornell University.
- Field Scouting Guide: Black Rot of Brassicas, Growing Produce, Meister Media Worldwide’s Horticulture Group.
- Managing Black Rot of Cabbage and other Crucifer Crops in Organic Farming Systems, eOrganic, eXtension Foundation (extension.org).
- Brassicas, Black Rot, UMass Extension vegetable Program Fact Sheet, Center for Agriculture, Food and the Environment, College of Natural Sciences, University of Massachusetts, Amherst, MA.
Club root of brassica vegetables
Disease: Club root of brassica vegetables
Pathogen: Plasmodiophora brassicae
Host crops: Broccoli, cabbage, cauliflower, brassicaceae (cruciferous) weeds, and radish.
On-Line Resources:
- Pacific Northwest Plant Disease Management Handbook: Cabbage and Cauliflower (Brassica sp.) – Clubroot.
- Clubroot of Crucifers. Vegetable MD Online.
- Clubroot. Wikipedia.
- Clubroot of vegetable brassicas – towards integrated control. New Zealand Institute for Crop & Food Research Ltd.
- Clubroot of Crucifers. The Ohio State University Extension.
- Managing Clubroot: Equipment Sanitation Guide (pdf). Canola Council of Canada.
- Top 10 tips from the 2013 International Clubroot Workshop. Canola Watch, Canola Council of Canada.
- See Diseases, pests, and other problems common to many vegetables: Clubroot of brassica vegetables.
White rust
Disease: White rust
Pathogen: Albugo candida
If you need the content provided on this site in an alternate format or for further information on the Vegetable Seed Pathology program,
please contact: Lindsey du Toit at dutoit@wsu.edu or at 360-848-6140.
Our pages provide links to external sites for the convenience of users. WSU Extension does not manage these external sites, nor does Extension review, control, or take responsibility for the content of these sites. These external sites do not implicitly or explicitly represent official positions and policies of WSU Extension.