Diseases Common to Brassica Vegetables
Insects/Mites
Abiotic Problems Common to Brassica Vegetables
Problems on Specific Brassicaceae Crops
(Click on photo to enlarge)
General Disease Management in Brassicas
- Production of Brassica Seed Crops in Washington State: A Case Study on the Complexities of Coexistence, Washington State University Extension Bulletin No. EM062E.
- Crucifer Disease Guide – A Practical Guide for Seedsmen, Growers and Agricultural Advisors. Published by Seminis Vegetable Seeds, Inc.’s Plant Health Department and Seed Health Departments.
Small-Scale Cost-Effective Hot Water Seed Treatment
By Frank Morton (Wild Garden Seed), Tom Stearns (High Mowing Seeds), Nick Andrews (OSU Small Farms Extension)
Hot water seed treatment is an important way of reducing the risk of seed-borne diseases, especially for organic Brassica growers (seed, fresh market or processed market) now that Pacific Northwest growers are dealing with black leg and potential light leaf spot epidemics. Hot water treatment can improve our defense against diseases like blackleg, light leaf spot, Verticillium, Fusarium, Xanthomonas, Alternaria, Botrytis and many seed-bourne viruses.
The authors have put together some slides with some practical ideas for low-cost and efficient hot water treatment.
Download the Hot Water Treatment Slideshow (pdf).
Diseases Common to Brassica Vegetables
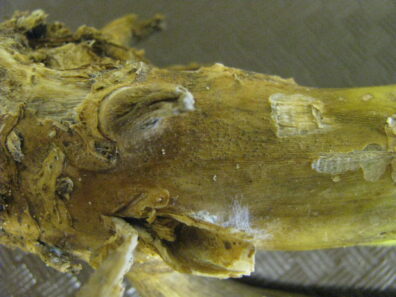
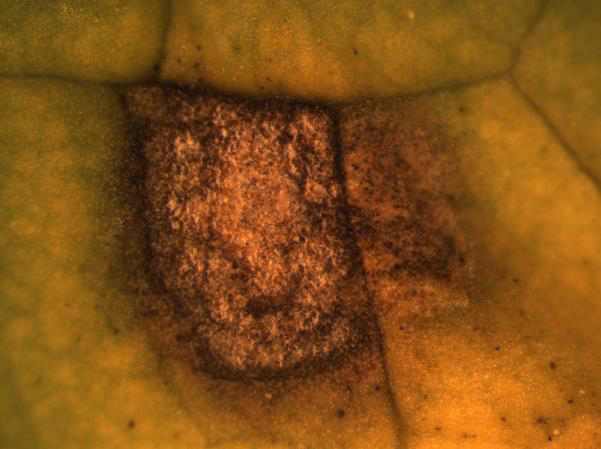
Black leg
Disease: Black leg
Pathogens: Phoma lingam (sexual stage = Leptosphaeria maculans)
Host crops: Most members of the Brassicaceae (Cruciferae) = cabbage family, including broccoli, Brussels sprouts, cabbage, canola, cauliflower, various Chinese brassica vegetables, collard, kale, mizuna, mustard, oilseed rape, oilseed turnip rape, rutabaga, turnip, etc.), Sinapis (white and yellow mustard), and Raphanus (daikon and radish). Several wild species exist that may be infected by P. lingam including Descurainia (tansymustard), Sisymbrium (hedge mustard), and Thlaspi (penny-cress). This is a quarantine disease in six counties in northwestern WA and all counties east of the Cascade Mountains because of the risk of this pathogen to the brassica vegetable seed industry.
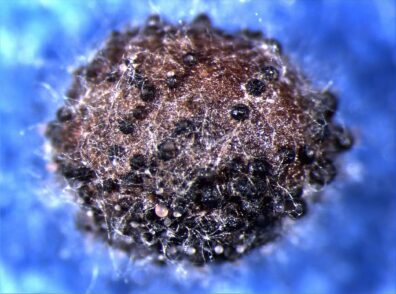
Photo Source: Lindsey du Toit, Washington State University
On-Line Resources:
- Video: Blackleg Disease and Resistance Management. Published by the Canola Council of Canada.
- Pacific Northwest Plant Disease Management Handbook: Cabbage and Cauliflower (Brassica sp.)-Black Leg and Phoma Root Rot.
- Pacific Northwest Plant Disease Management Handbook: Seed Crop, Crucifers (Brassica and Raphanus spp.)-Black Leg.
- Black leg in Brassicaceae crops and wild crucifers: 2014 outbreak in the Willamette Valley of Oregon (pdf).
- Black Leg, Light Leaf Spot, and White Leaf Spot (pdf), Cynthia Ocamb, PhD., Plant Pathologist, OSU Extension, Associate Professor–Botany & Plant Pathology.
- Fungicides for Control of Black Leg (pdf), David Priebe, Pesticides Program, Oregon Department of Agriculture.
Addressing Blackleg in the Willamette Valley: Oregon Department of Agriculture permanent ruling released on black leg of brassicaceae in January 2015 – see the Brassica Production Districts document, and the OSDA Permanent Ruling document titled ‘Crucifer blackleg disease requirements moved into one regulation; removes same requirements from rapeseed production districts,’ below.
- Oregon Secretary of State Certificate and Order for Filing (pdf) – PERMANENT ADMINISTRATIVE RULES: Crucifer blackleg disease requirements moved into one regulation; removes same requirements from rapeseed production districts.
- Brassicaceae Production Districts and Rapeseed Control Areas (pdf) (603-052-0860), Oregon Department of Agriculture.
- Management of Black Leg in Oregon on Brassica seed crops (pdf), a Clinic Close-up, Oregon State University Extension Service.
- Management of Black Leg in Oregon on Vegetable Brassica Crops and Seed Crops (pdf), a Clinic Close-up, Oregon State University Extension Service.
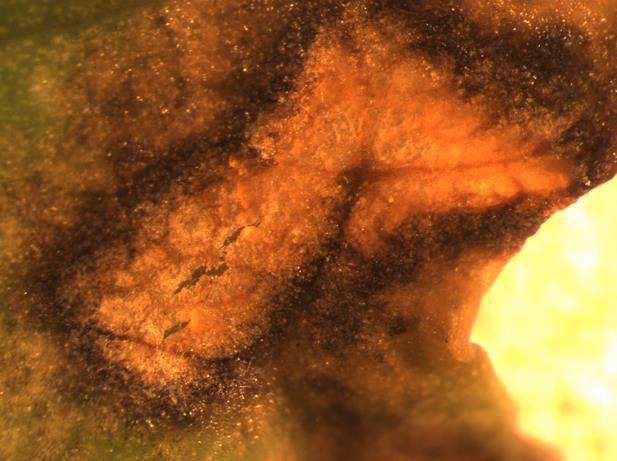
Black rot
Disease: Black rot
Pathogens: Xanthomonas campestris pv. campestris
Host crops: Most members of the Brassicaceae (Cruciferae) = cabbage family, including broccoli, Brussels sprouts, cabbage, canola, cauliflower, various Chinese brassica vegetables, collard, kale, mizuna, mustard, oilseed rape, oilseed turnip rape, rutabaga, turnip, etc.), Sinapis (white and yellow mustard), and Raphanus (daikon and radish). Most wild species can be infected by this pathogen. This is a quarantine disease in parts of six counties in northwestern Washington because of the risk of this pathogen to the brassica vegetable seed industry.
Online Resources:
- Cabbage and Cauliflower (Brassica sp.)-Black Rot, Pacific Northwest Handbooks, a Pacific Northwest Extension Publication.
- Black Rot of Crucifers, Fact Sheet, Cooperative Extension, New York State, Cornell University.
- Field Scouting Guide: Black Rot of Brassicas, Growing Produce, Meister Media Worldwide’s Horticulture Group.
- Managing Black Rot of Cabbage and other Crucifer Crops in Organic Farming Systems, eOrganic, eXtension Foundation (extension.org).
- Brassicas, Black Rot, UMass Extension vegetable Program Fact Sheet, Center for Agriculture, Food and the Environment, College of Natural Sciences, University of Massachusetts, Amherst, MA.
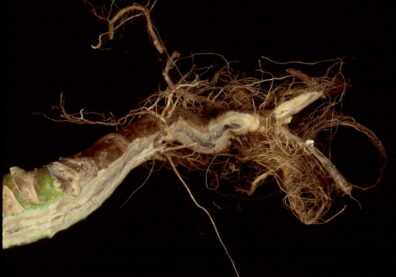
Clubroot
Disease: Clubroot
Pathogen: Plasmodiophora brassicae
Host crops: Broccoli, cabbage, cauliflower, brassicaceae (cruciferous) weeds, and radish.

Photo Source: Lindsey du Toit
On-Line Resources:
- Pacific Northwest Plant Disease Management Handbook: Cabbage and Cauliflower (Brassica sp.)-Clubroot.
- Clubroot of Crucifers. Vegetable MD Online.
- Clubroot. Wikipedia.
- Clubroot of vegetable brassicas – towards integrated control. New Zealand Institute for Crop & Food Research Ltd.
- Clubroot of Crucifers. The Ohio State University Extension.
- Managing Clubroot: Equipment Sanitation Guide (pdf). Canola Council of Canada
- Top 10 tips from the 2013 International Clubroot Workshop. Canola Watch, Canola Council of Canada.
- See Diseases, pests, and other problems common to many vegetables: Clubroot of brassica vegetables.
Downy mildew
Disease: Downy mildew
Pathogens: Hyaloperonospora parasitica = Peronospora parasitica
Host crops: Most members of the Brassicaceae (Cruciferae) = cabbage family, including cabbage and cauliflower.
Online Resources:
- Pacific Northwest Plant Disease Management Handbook: Cabbage and Cauliflower (Brassica sp.) – Downy Mildew {Staghead}.
- Pacific Northwest Plant Disease Management Handbook: Mustard Greens (Brassica juncea) – Downy Mildew {Staghead}.
- Downy Mildew of Crucifers (pdf). Plant Pathology Fact Sheet. Florida Cooperative Extension Service.
- Brassica Downy Mildew. University of Massachusetts Amherst.
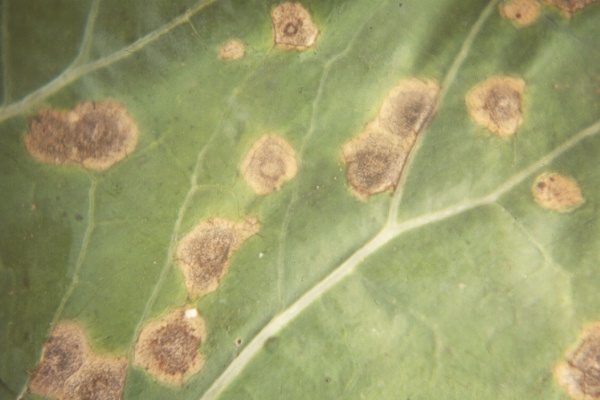
Light leaf spot
Disease: Light leaf spot
Pathogens: Cylindrosporium concentricum (sexual stage: Pyrenopeziza brassicae)
Host crops: Light leaf spot has been observed causing disease in canola (oilseed rape can be very susceptible), forage Brassica species, “field” turnip, other Brassica members including wild mustard, volunteer black mustard, vegetable Brassica seed fields, and Brassica species used as cover crops. It is likely that all brassicas crops grown in the Pacific Northwest are susceptible with a range of susceptibility within each crop species.
Online Resources:
Ring spot
Disease: Ring spot
Pathogen: Mycosphaerella brassicicola
On-Line Resources:
White leaf spot
Disease: White leaf spot
Pathogens: Pseudocercosporella capsellae (sexual stage: Mycosphaerella capsellae)
Host crops: White leaf spot and gray stem were observed during 2014 in canola research fields as well as in commercial seed fields of forage Brassicas and “field” turnip. White leaf spot was also detected in volunteer black mustard and forage fields. Susceptible hosts reportedly include species of Brassica (broccoli, cabbage, canola, cauliflower, Chinese cabbage, mustard, turnip, etc.) as well as radish and horseradish. Weedy types such as wild radish, wild mustard, and shepherd’s purse are susceptible to white leaf spot and gray stem.
Online Resources:
White mold
Disease: White mold
Pathogens: Sclerotinia sclerotiorum
Host crops: Most members of the Brassicaceae (Cruciferae = cabbage family) and many other vegetables (see Diseases, pests, and other problems common to many vegetables: White mold)
Online Resources:
- Management of Sclerotinia in Turnip Seed Crops (pdf) – A Clinic Close-up, Oregon State University Extension Service
- Pacific Northwest Plant Disease Management Handbook: Cabbage and Cauliflower (Brassica sp.) – Sclerotinia Stem Rot and Watery Soft Rot
- See Diseases, pests, and other problems common to many vegetables: White mold.
Insects/Mites
Root maggots
Common name: Root maggots (cabbage maggot and seedcorn maggot)
Latin binomial: Delia brassicae = Delia radicum (cabbage maggot), and Delia platura (seedcorn maggot)
Host crops: Cabbage maggot can damage and destroy root systems of all cole (crucifer or Brassicaceae) crops. Tunnels from the feeding maggots can become numerous in roots in crops with severe infestations. The tunnels provide wound sites for pathogens, particularly bacteria that can cause bacterial soft rot. The seedcorn maggot can affect many vegetable crops including snap, kidney, and lima beans, onion, corn, turnip, pea, cabbage, and cucurbits. They cause the most damage in spring to newly emerging seedlings, and can cause severe losses in plant stand.

Photo Source: Lindsey du Toit, Washington State University
Online Resources:
Abiotic Problems Common to Brassica Vegetables
Boron (B) deficiency
Problem: Boron (B) deficiency
Crops affected: Most crops can develop symptoms of boron (B) deficiency. Brassica or cole crops have moderate to high B requirements. B deficient cole crops can develop cracked, corky stems, as well as petioles and midribs. Broccoli, cabbage and cauliflower stems may become hollow and discolored. Cauliflower curds may turn brown and leaves roll and curl. Cabbage heads may be smaller than normal and discolored yellow. Cauliflower is the most sensitive of cole crops to B deficiency.
Online Resources:
- Boron Deficiencies in Cole Crops, University of Delaware Extension.
- Boron Deficiency
- Boron
- Boron mobility in plants (pdf). Chapter 7 from the book Plant and Soil by Patrick H. Brown, Department of Pomology, University of California, Davis and Barry J. Shelp, Department of Horticultural Science, University of Guelph, Guelph, Ontario.
- Boron Deficiency Symptoms (pdf) U.S. Borax Corp.
- Boron in vegetables (pdf) U.S. Borax Corp.
Edema
Problem: Edema
Cause: A physiological problem prominent when air is cooler than the soil, soil moisture is high, and relative humidity is high. The low plant transpiration rates combined with an increase in water absorption by roots from the soil leads to increased cell turgor pressure, resulting in eruption of epidermal cells as the inner cells enlarge. Protrusion of the inner cells causes epidermal cells to die and discolor, resulting in a ’warty’ appearance that can be misidentified as a disease. Symptoms are usually worse on the lower leaf surface and on older (lower) leaves.
Host Crops: Numerous vegetables including spinach, brassicas, tomato, etc. Vegetables with waxy leaves, e.g., brassicas, tend to be most susceptible.
Online Resources:
- Pacific Northwest Handbooks: Cabbage and Cauliflower Brassica SP Oedema Edema.
- What are these bumps on my vegetables? Edema or oedema: It doesn’t matter how you spell it, it still doesn’t look good. What is it, what causes it and how can I prevent it? Michigan State University Extension.
Redheart
Problem: Redheart
Cause: Freeze damage to inner leaves of a brassica head as a result of an extended period (>24 hours) of freezing in the field or in storage. Damage is often irreversible. Leaves several layers inside the head become watersoaked/glassy while outer leaves of the head appear normal. Internal affected leaves become tan or red in color, and may dry to a papery texture. A bad odor may develop. A dark zone may delimit the affected vs. healthy areas. Similar symptoms can be caused by exposure of cabbage heads to low oxygen levels or high carbon dioxide levels in controlled atmosphere storage.
Crops affected: Brassica vegetable crops with heads, e.g., cabbage. Some cultivars are more sensitive to redheart than others.
Harvest crops before extended periods of severe frost. If freezing conditions occur, allow heads to thaw completely before they are harvested. Monitor the heads at harvest for glassiness of the internal leaves. For longer-term storage, only store cabbage heads that have not been exposed to frost. Ventilate storage rooms adequately at temperatures just above freezing and high humidity.
Harvest crops before extended periods of severe frost. If freezing conditions occur, allow heads to thaw completely before they are harvested. Monitor the heads at harvest for glassiness of the internal leaves. For longer-term storage, only store cabbage heads that have not been exposed to frost. Ventilate storage rooms adequately at temperatures just above freezing and high humidity.
Online Resources:
Stem splitting
Problem: Stem splitting
Crops affected: Any brassica crop grown for seed can develop stem splitting under conditions that promote very rapid growth (high soil moisture and warm temperatures).

Photo Source: Karen Sowers, Washington State University Dept. of Crop & Soil Sciences

Photo Source: Karen Sowers, Washington State University Dept. of Crop & Soil Sciences

Photo Source: Karen Sowers, Washington State University Dept. of Crop & Soil Sciences

Photo Source: Karen Sowers, Washington State University Dept. of Crop & Soil Sciences
If you need the content provided on this site in an alternate format or for further information on the Vegetable Seed Pathology program,
please contact: Lindsey du Toit at dutoit@wsu.edu or at 360-848-6140.
Our pages provide links to external sites for the convenience of users. WSU Extension does not manage these external sites, nor does Extension review, control, or take responsibility for the content of these sites. These external sites do not implicitly or explicitly represent official positions and policies of WSU Extension.