Newsletter Archives |
August 2003 Newsletter
Lindsey du Toit and Debra Inglis, editorsWSU Mount Vernon NWREC
16650 State Rte 536, Mount Vernon, WA 98273-4768
360-848-6140 (tel), 360-848-6159 (fax)
WSU Vegetable Pathology Team Newsletter
IN THIS ISSUE
- Hello
- Pest Alert (High Plains Virus on Sweet Corn)
- Upcoming Vegetable Events and Announcements
- Information About Diagnosing Leafy Vegetable Diseases
HELLO!
Welcome to the August 2003 edition of Washington State University's Vegetable Pathology Extension Team newsletter, the final edition of the 2003 growing season. If you have suggestions for next year's growing season newsletters, please let us know.
If you havequestions or comments about vegetable diseases and pests, or WSU's Vegetable Pathology Team, contact Debbie Inglis (dainglis@wsu.edu) or Lindsey du Toit (dutoit@wsu.edu).
PEST ALERT!!!
Field Days
High Plains Disease Caused by the High Plains Virus
Photos courtesy of Gary Q. Pelter
There
have been reports this season of severe outbreaks of high plains
disease (HPD) in sweet corn fields in several counties in the
Columbia Basin. HPD is caused by the High plains tenuivirus
(HPV). The disease was first detected in Colorado, Kansas, and
Texas in 1993; in Utah in 1994; and in Washington in 1998. |
|
|
Symptoms: Mosaic symptoms are usually first evident on leaves emerging from the whorl, although older leaves may also develop mosaic symptoms (Fig. 1 and 2). Infected plants may be stunted and yellowish (Fig. 3). Yellow or red-purple (depending on the genetic background of the corn cultivar) bands ¼ to ½" in width and running parallel to the veins may be observed on intermediate to fully-developed leaves. These bands later turn necrotic. Infected plants may have stunted or weakened root systems. |
Transmission: HPV is transmitted by: 1) the wheat curl mite
(Aceria tosichella), a tiny eriophyid mite which also
transmits wheat streak mosaic virus (WSMV); and 2) corn
seed. Corn plants can be infected with both HPV and WSMV. The
mite, which is barely visible with a 10x magnifying lens, can
be blown between fields. As a result, a distinct disease gradient
is sometimes evident on corn crops planted immediately adjacent
to infected wheat crops. Seed transmission is thought to be
very low, although the pattern of symptom development in some
outbreaks of HPD in sweet corn crops have suggested higher incidences
of seed transmission. |
|
Management: Management options for HPD are limited. Avoid seeding crops late. Control grassy weeds and volunteer wheat, which may serve as reservoirs of HPV and on which the wheat curl mite may persist. If possible, avoid planting sweet corn crops immediately adjacent to wheat crops. The objective is to break the 'green bridge' effect and prevent spread of the HPV in the mite vector. As resistance screening efforts progress, plant resistant corn cultivars. Limited information is currently available on the resistance of sweet corn hybrids to HPD, but known susceptible hybrids include: Ambrosia, Challenger, Crisp 'n Sweet, DMC 20-3, DMC 20-10, Double Gem, Extra Sweet, Honey and Pearl, How Sweet It Is, Native Gem, Phenominal, Shasta, Style Sweet, and 710. Resistant or tolerant hybrids include: Delectable, Empire, Gemini, Imaculata, Incredible, Platinum Lady, Silver Queen, and 711.
UPCOMING VEGETABLE EVENTS AND ANNOUNCEMENTS
New Book on Genetically Modified Crops
The World Scientific Publishing of the Imperial College Press in London has published "Genetically Modified Crops" by Nigel G. Halford. The book describes the history and development of the science of biotechnology. It also features the GM crops that are grown commercially around the world, and the new varieties that are currently being developed. More information can be found at https://www.wspc.com.sg/books/lifesci/p284.html
New Bulletin from Skagit Veg Trials Available
A new bulletin "Temperature Modification Techniques for the Growing Environment in the Puget Sound" prepared by the volunteers of the Skagit Veg Trials and Dr. Andy Anderson, Skagit Veg Trials Director, is now available. Information on transplanting, raised beds, solar mulch, cloches, plastic mulches and combined strategies for modifying soil temperature is included. For copies, contact andersonw@wsu.edu
New Organic Agriculture Resource Guide
OrganicAgInfo is an on-line database of research reports, farmer-to-farmer information, and outreach publications on organic agriculture. The database is being hosted by North Carolina State University, and was funded by grants to the Scientific Congress on Organic Agricultural Research and the Organic Agriculture Consortium from the Initiative for Future Agriculture and Food Systems through USDA-CSREES. See https://www.organicaginfo.org
Onion Field Day
The 2003 Columbia Basin Onion Field Day will be held Thursday, August 28 from 9:00 am until noon at Grigg and Sons Farm near Quincy, WA. Forty-six onion cultivars and lines from nine seed companies will be compared and contrasted, and gray mold research explained. For more information, contact Gary Pelter at 509-754-2011.
Plant Disease Diagnosis
For a comprehensive guide to plant disease diangosis see the APSnet Education Center Introductory Topic article written by M. B. Riley, M. R. Williamson and O. C. Maloy. It can be viewed at https://www.apsnet.org/education/IntroPlantPath/Topics/Default.htm
Searching for Plant Science Images?
Check the Plant Management Network Image Collections at https://www.plantmanagementnetwork.org/images/ The site contains more than 1,500 high quality photographs, graphics, charts, graphs, maps, biological drawings, chemical structures, diagrams and disease cycles focused on plant science. Subscriptions are $38. See https://www.plantmanagementnetwork.org/about/subscribe/
WSU Cooperative Extension Has On-line Publications
You
can download hundreds of WSU publications free of charge at the
new WSU publications catalog and sales site at https://pubs.wsu.edu/.
Should you choose others, ordering is very easy. Temporary discounts
of up to 25% on some publications are being offered.
NFORMATION ABOUT DIAGNOSING LEAFY VEGETABLE DISEASES
Foliar diseases on leafy vegetables in northwestern Washington
The following is an excerpt from the WSU-Vegetable Pathology Team's Leafy Vegetable Workshop, presented by Lindsey du Toit, Jenny Glass and Debbie Inglis on July 24, 2003 at WSU-Mount Vernon.
Foliar diseases on leafy vegetable crops in Washington can be caused by both obligate and non-obligate parasites. Obligate parasites are organisms that can grow and multiply only in or on living tissue, and include Peronospora, an oomycete causing downy mildew; Erysiphe, one of the fungi causing powdery mildew; Albugo, an oomycete causing white rust; and, Uromyces and Puccinia two fungi which cause common rust. Erisphe belongs to the ascomycetes, and Uromyces and Puccinia are basidiomycetes. Some examples of non-obligate parasites include spinach leaf spot fungi (Cladosporium, Stemphylium, and Colletotrichum); the beet and chard leaf spot fungi (Phoma, Ramularia, and Cercospora), and crucifer foliar pathogens (Alternaria and Mycosphaerella). These fungi are ascomycetes or deuteromycetes.
Correct diagnoses of leafy vegetable foliar diseases are important because they can lead to appropriate fungicide selection, improve the timing of fungicide applications, assist with the selection of cultivars with higher levels of resistance, and determine appropriate cultural practices for more effective disease management.
The downy mildews are some of the most commonly encountered foliar diseases on leafy vegetables in the PNW. They are also relatively easy to diagnose (see photos in table below). Favorable conditions for downy mildew outbreaks include cool temperatures ranging from 50 to 60 °F and high moisture related to low cloud ceilings, dew, fog and rain.
Downy mildews on leafy vegetables.
|
Symptoms |
Disease and pathogen |
Comments |
|
|
Peronospora farinosa f. sp. spinaciae on spinach |
Note chlorosis on upper leaf surface, and grayish brown felt-like mildew growth on leaf undersurface |
|
|
Peronospora farinosa f. sp. betae on Swiss chard |
Note grayish green felt-like growth and twisted leaf |
|
|
Peronospora farinosa f. sp. betae on beet |
Note pinkish felt-like growth on leaves and twisted seed stalk |
|
|
Peronospora parasitica on cabbage |
Note chlorotic spots on upper leaf surface, and grayish white crust-like growth on leaf undersurface |
|
|
Peronospora parasitica on broccoli |
Note dark streaking in vascular tissue due to systemic infection |
Leaf spot diseases on leafy vegetables are quite common in the PNW, although they can be somewhat more challenging to diagnose than downy mildews. Often the pathogens need to be cultured from the diseased leaf tissues, and the spores from the fungal colonies identified with the use of a microscope before the diagnosis can be made.
Three fungal leaf spot diseases of spinach.
|
|
Cladosporium leaf spot |
Stemphylium leaf spot |
Anthracnose |
|
Pathogen name |
Cladosporium variabile |
Stemphylium botryosum |
Colletotrichum dematium |
|
Leaf spot symptoms |
Distinct, 1-3 mm spots, develop dark margin |
Diffuse, rapidly expanding |
Distinct, coalescing, water-soaked in humid conditions |
|
Spores in lesions |
+ |
+ |
+ |
|
Dispersal |
Wind, seed |
Wind, seed |
Splashing water, seed |
|
Overwintering |
Volunteers, seed |
Woody spinach debris, seed |
Volunteers, seed |
|
Favorable conditions |
Moist, cool |
Moist, warm, pollen on leaves |
Wet, cool |
|
Host range |
Chenopod species, others? |
Spinach |
Spinach |
 |
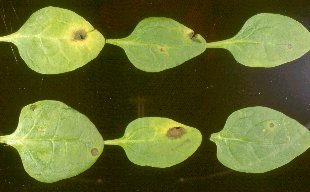 |
Fig. 1. Cladosporium leaf spot (left) and Stemphylium leaf spot (right) on spinach. Photo courtesy of Mike L. Derie. |
Fig. 2. Anthracnose on young spinach leaves caused by Colletotrichum dematium. Photo courtesy of Mike L. Derie. |
Three fungal leaf spot diseases of beets and chard.
|
Cercospora leaf spot |
Ramularia leaf spot |
Phoma leaf spot |
|
|
Pathogen name |
Cercospora beticola |
Ramularia betae |
Phoma betae |
|
Symptoms |
Circular leaf spots, red-brown margin, on older leaves; Also causes crown lesions |
Light brown leaf spots, angular & larger, on older leaves |
Round leaf spots, concentric rings on perimeter, dark margin; Also causes seedling black leg & crown rot |
|
Spores in leaf spots |
Minute black dots (stromata) |
Silvery gray to white spore masses |
Black fruiting bodies (pycnidia), also in crown lesions |
|
Seedborne |
+ (external) |
+ (?) |
+ |
|
Dispersal |
Splashing water, wind, insects, seed |
Wind, seed |
Splashing water, insects, seed |
|
Overwintering |
Weeds, debris, seed |
Debris, seed |
Soil, roots, debris, weeds, seed |
|
Favorable conditions |
Warm, moist |
Cool, moist |
Cool to warm, moist |
|
Host range |
Beet, chard, Chenopod. weeds |
Beet, chard |
Beet, lambsquarter |
 |
 |
 |
Fig.
1. Cercospora leaf spot on sugar beet. |
Fig. 2. Ramularia leaf spot of chard. Photo courtesy of Debra A. Inglis. |
Fig. 3. Phoma leaf spot on beets. Photo courtesy of Lindsey J. du Toit. |
Two fungal leaf spot diseases of crucifer crops.
|
Alternaria leaf & pod spot |
Ring spot |
|
|
Pathogen name |
Alternaria
brassicicola & |
Mycosphaerella brassicicola |
|
Symptoms |
Black circular to irregular lesions, necrotic center; Circular to elongated black spots on pods and racemes |
Circular lesions (up to 2 cm diameter), definite margin & chlorotic halo, concentric zonation, on older leaves |
|
Spores in leaf spots |
Naked spores |
Black fruiting bodies (pycnidia &/or perithecia) in concentric rings |
|
Seedborne |
+ |
- |
|
Dispersal |
Wind, insects, windblown rain |
Splashing water, insects |
|
Overwintering |
Debris, biennial seed crops, seed |
Debris, biennial seed crops |
|
Favorable conditions |
Moist, warm |
Moist, cool |
|
Host range |
Crucifers |
Crucifers |
 |
 |
Fig. 1. Alternaria leaf and pod spot caused by A. brassicicola & A. brassicae. Photos courtesy of Lindsey J. du Toit. |
Fig. 2. Ring spot of cabbage caused by Mycosphaerella brassicicola. Photo courtesy of Lindsey J. du Toit. |
Bacterial foliar diseases of crucifer crops.
|
Black rot |
Xanthomonas leaf spot |
Peppery leaf spot |
||
|
Pathogen name |
Xanthomonas campestris pv. campestris |
X. campestris pv. raphani |
X. campestris pv. armoraciae |
Pseudomonas syringae pv. maculicola |
|
Symptoms |
Yellow leaves, wilting, black veins, V-shaped lesions from leaf margin |
Circular, water-soaked leaf spots, chlorotic halo; dark lesions on petioles |
Circular to angular spots, chlorotic halo |
|
|
Systemic |
+ |
- |
- |
|
|
Seedborne |
+ |
+ |
+ |
+ |
|
Dispersal |
Splashing water, seed, insects |
|||
|
Overwintering |
Debris, cruciferous weeds, soil |
|||
|
Favorable conditions |
Warm to hot, moist |
Cool to warm, extended wet periods |
Cool, wet |
|
|
Host range |
Crucifers (including weeds) |
Cabbage, broccoli, cauliflower, kale, radish, tomato, pepper |
Cabbage, broccoli, cauliflower, kale, radish, horseradish |
Cabbage, broccoli, cauliflower, Brussel sprouts, turnip |
 |
 |
Fig. 1. Black rot of cabbage showing blackened veins, V-shaped marginal lesions, chlorosis and wilting. Photo courtesy of Lindsey J. du Toit. |
Fig. 2. Xanthomonas leaf spot of crucifers caused by X. campestris pv. raphani & X. campestris pv. armoraciae. Photo courtesy of Lindsey J. du Toit. |
Our pages provide links to external sites for the convenience of users. WSU Extension does not manage these external sites, nor does Extension review, control, or take responsibility for the content of these sites. These external sites do not implicitly or explicitly represent official positions and policies of WSU Extension.